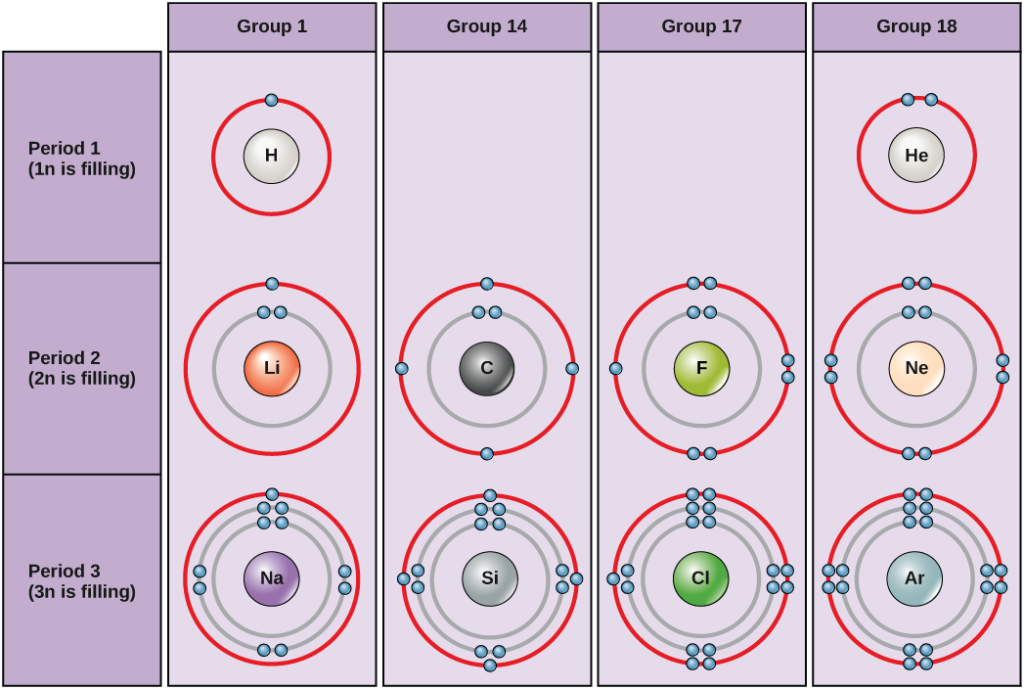
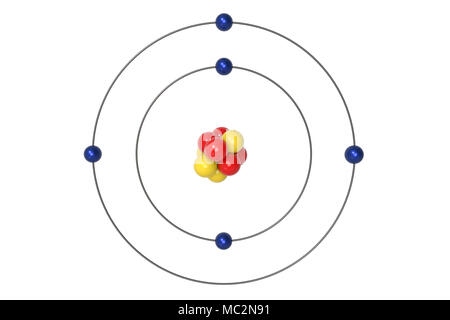
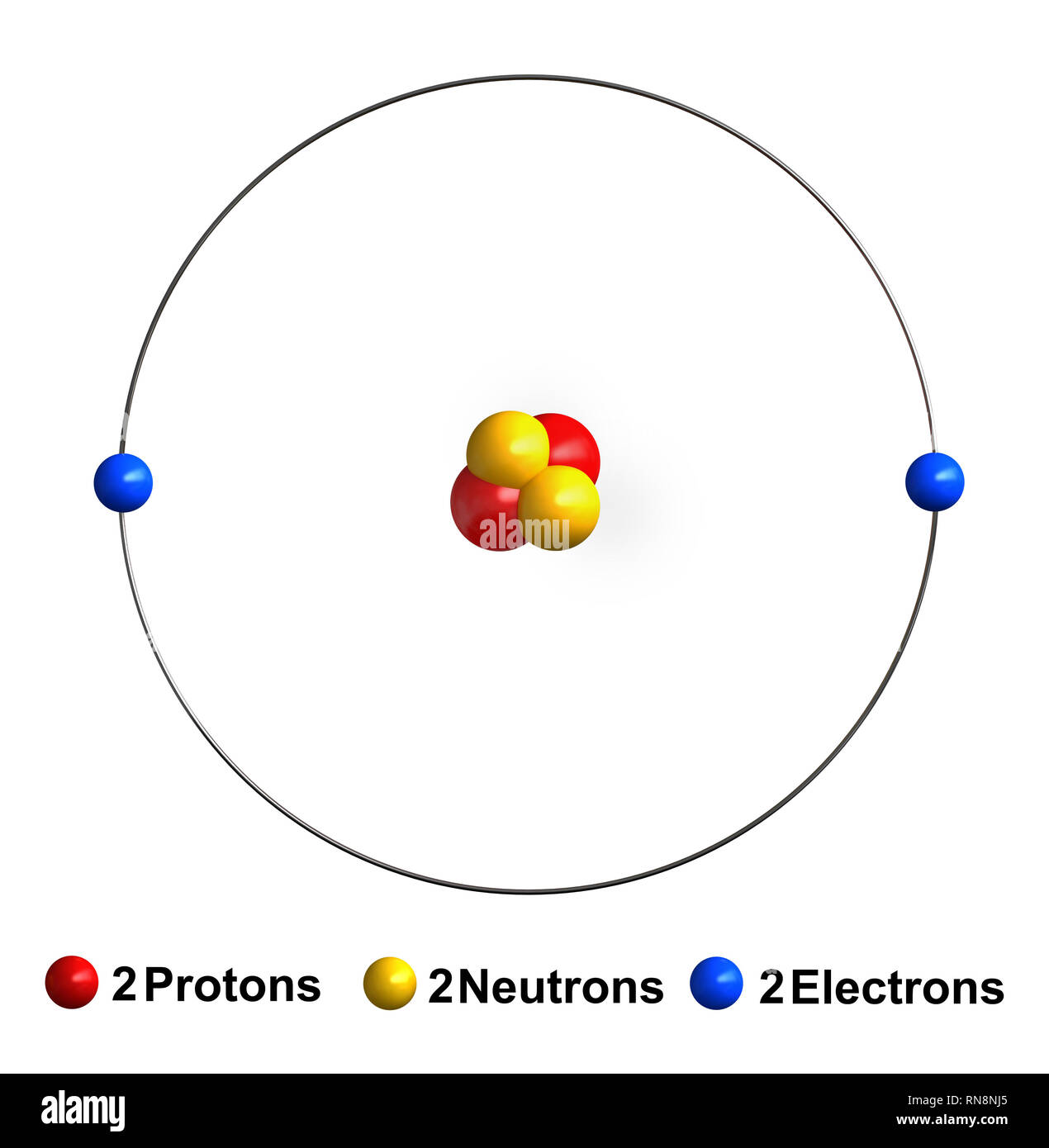
 Solve the equation after substituting the values in the formula. The simple formula to calculate the Bohr's radius is r = (4πϵ 0. The amount of energy absorbed or emitted can be calculated using the orbital energies difference. Every orbit has an energy associated with it. Energy is emitted when the electron jumps from higher to lower orders and absorbed when jumps from a lower orbit to the higher. Those orbits are called stationary orbits. The four important principles of the Bohr model are electrons occupy only certain orbits around the nucleus. What are the 4 principles of Bohr's model?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
